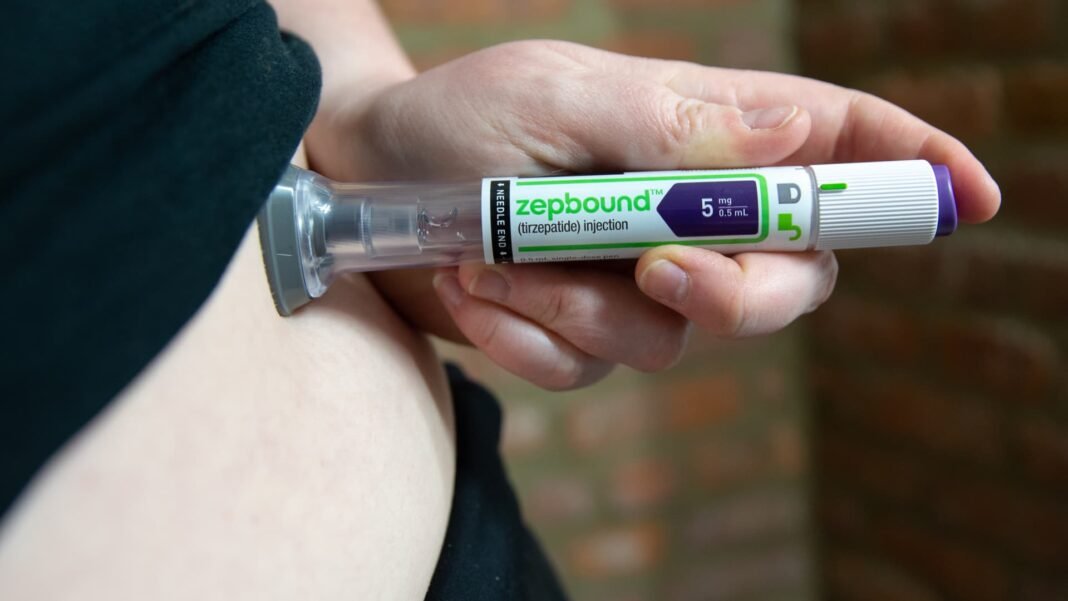
Revolutionizing Obesity Care: The Launch of the Multi-Dose Zepbound KwikPen
Eli Lilly & Co. has introduced a groundbreaking delivery method for itS popular obesity medication, Zepbound, by launching a single pen that contains an entire month’s worth of doses. This innovative device, called the KwikPen, consolidates four weekly injections into one user-friendly pen designed to improve treatment convenience and adherence.
Enhancing Patient Experience with the KwikPen
The new KwikPen offers a streamlined alternative to traditional weekly single-dose auto-injectors. Instead of juggling multiple pens each month, patients can now rely on one multi-dose device that delivers all four required injections over 28 days. This simplification is expected to boost compliance and reduce the complexity frequently enough associated with chronic obesity management.
For individuals covering costs out-of-pocket, the multi-dose KwikPen is accessible through Lilly’s direct-to-consumer platform at an entry price of $299 per month for the lowest dosage tier.
Zepbound’s rapid Growth and Market Influence
As its introduction in late 2023, Zepbound has witnessed remarkable demand growth across U.S. markets. Its rapid adoption has positioned eli Lilly as a leading force in weight loss pharmaceuticals, overtaking competitors like Novo Nordisk in market share.
In just one quarter following launch, Zepbound generated $4.2 billion in revenue within the United States-a staggering 122% increase compared to prior year figures-demonstrating strong patient preference for effective obesity treatments amid rising global health concerns related to excess weight.
The Impact of Direct-to-Consumer Sales channels
LillyDirect-the company’s dedicated online sales portal-has been pivotal in expanding access to Zepbound and its new multi-dose format. By initially offering this advanced delivery system exclusively through this channel, Eli Lilly aims to attract users who prioritize ease-of-use and integrated medication management solutions.
Regulatory Endorsement and Device Provenance
The Food and Drug Management (FDA) recently approved an expanded indication for Zepbound that includes authorization for use with the multi-dose KwikPen injection device-affirming both safety standards and therapeutic effectiveness.
This type of delivery mechanism is not unfamiliar; it mirrors technology already employed successfully by other Eli Lilly products such as Mounjaro®, their widely trusted diabetes injection pen used globally by millions managing type 2 diabetes daily.