Manitoba Patient Faces Imminent Loss of Vital Therapy for progressive Neuromuscular Disorder
A Manitoba resident living with a debilitating condition that severely impairs muscle function now confronts the threat of losing access to a medication that has significantly improved his quality of life and slowed disease progression.
Critical Medication Coverage at Risk
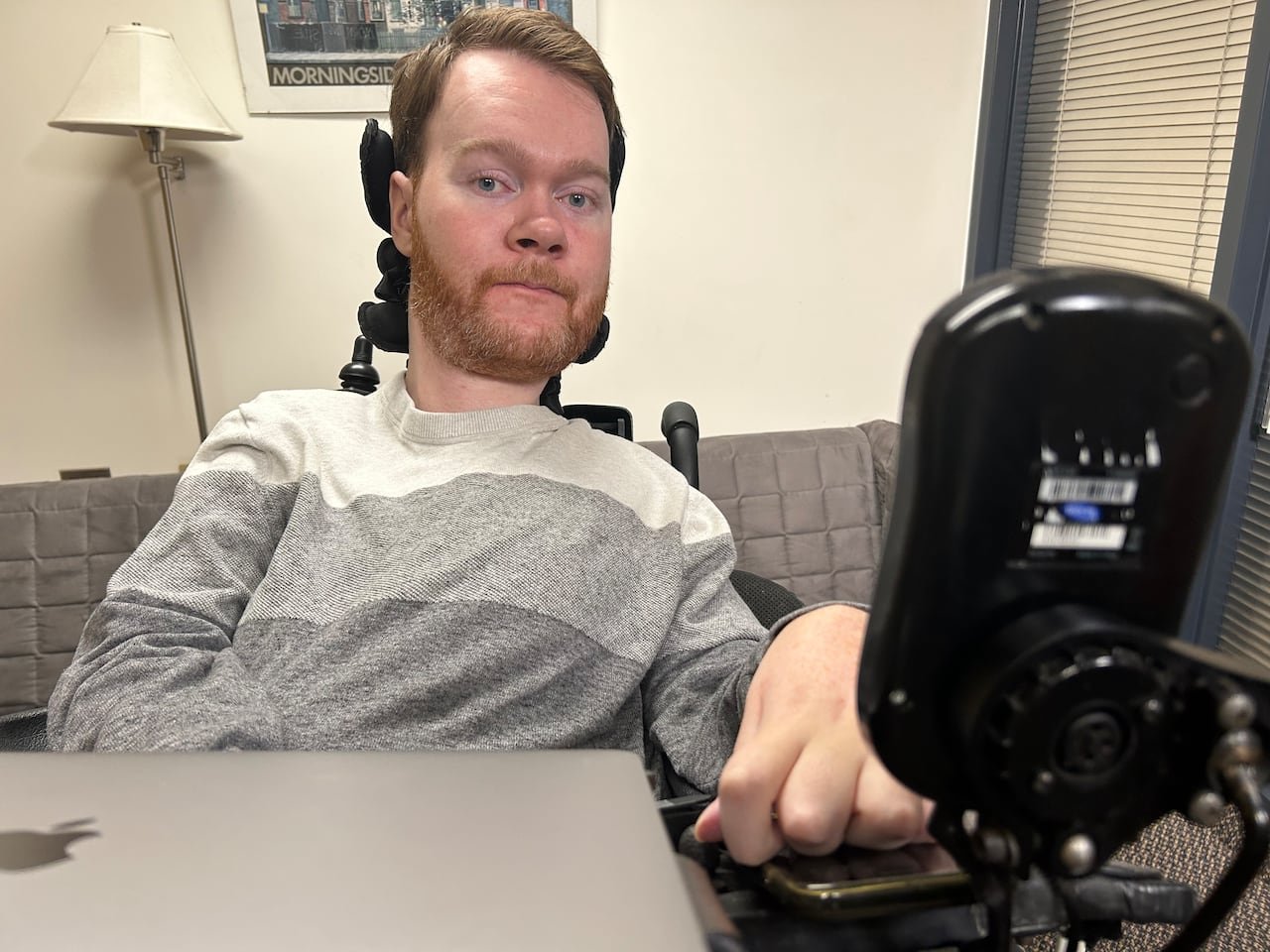
Jeremy Bray, aged 30 from Rivers, Manitoba, is diagnosed with Type 2 spinal muscular atrophy (SMA), a rare inherited disorder characterized by gradual muscle degeneration. Since beginning treatment in May with risdiplam (Evrysdi)-a revolutionary drug provided free through Roche’s compassionate use program-Jeremy has seen remarkable gains. His facial expressions have become more vivid, his voice stronger, and he can work longer hours without exhaustion.
Now facing the imminent end of this free supply as Roche discontinues its no-cost provision, Jeremy urgently appeals to Manitoba’s government to step in and fund this essential therapy before further deterioration strips away his remaining independence.
The Challenge of SMA and Barriers to Treatment Access
SMA progressively diminishes voluntary muscle control; Jeremy currently depends on limited facial muscles and one thumb for communication due to loss of limb and head mobility. The introduction of risdiplam offered an unprecedented reversal in what was previously an inevitable decline.
“I never imagined my condition could improve,” Jeremy reflects. “This medication gave me hope when I thought all was lost.”
Provincial Restrictions Limit Adult Access Despite Positive Outcomes
The manitoba government restricts funding for risdiplam exclusively to patients under 25 years old based on recommendations from canada’s Drug Agency (CDA). The agency argues there is insufficient clinical trial data involving adults over 25 years old-leaving older patients like Jeremy without coverage despite demonstrated benefits.
This policy contrasts sharply with provinces such as Quebec-which imposes no age restrictions-and others including Saskatchewan, Alberta, and Ontario that have occasionally approved adult treatments based on individual assessments despite CDA guidelines.
Growing Evidence Spurs Calls for Policy Reassessment
This year Manitoba requested an expedited review by the CDA after neurologists presented real-world data showing meaningful improvements among adult SMA patients treated with risdiplam locally. However, the agency maintained its position citing lack of randomized controlled trials involving older adults-a global challenge since withholding effective therapies once proven is considered unethical in clinical research settings.
Diverse Provincial Funding Models Highlight Systemic Challenges
- Quebec: No age-based restrictions; regional clinicians report nearly 90% efficacy among adult SMA patients receiving risdiplam treatment.
- Saskatchewan & Alberta: Provide selective funding for adults following case-by-case evaluations despite federal recommendations against broad approval.
- manitoba: Enforces strict age limits aligned with CDA advice; denies funding requests even amid patient advocacy campaigns seeking exceptions.
- Ontario: Has granted coverage for some adult cases through special access programs reflecting increasing recognition of unmet needs within older SMA populations.
The Human Impact Behind Policy Decisions
“Consider if your child showed progress only to have it abruptly halted as bureaucratic rules prevent continued treatment,” stated Progressive Conservative leader Obby Khan. “Other provinces acknowledge this drug’s transformative potential-Manitoba must follow suit.”
The Manufacturer’s Outlook: Balancing Compassionate care with Sustainability
A representative from Roche highlighted that while compassionate use initiatives provide crucial short-term support for individuals like Jeremy Bray, these programs cannot substitute complete healthcare policies ensuring equitable access nationwide. maintaining indefinite free distribution is financially unsustainable and risks creating inequities between those who recieve assistance versus those excluded due to cost or policy constraints.
Pursuing Lasting Funding Frameworks
The company urges provincial health authorities to develop innovative reimbursement models enabling fair access across all eligible Canadians so transformative treatments are not dependent solely on manufacturer generosity or temporary measures but integrated into long-term healthcare strategies.
A Future Filled With hope Amid Ongoing uncertainty
SMA affects approximately one out of every 10,000 live births worldwide yet remains underfunded compared with other neurological diseases-even as recent therapeutic advances offer new hope where none existed before. As governments weigh financial costs against mounting evidence supporting expanded eligibility beyond traditional trial populations-patients like Jeremy face uncertain futures hinging on policy changes still pending today.
- total annual expenses per patient initiating risdiplam average around $300,000 CAD during the first year depending on dosage adjustments;